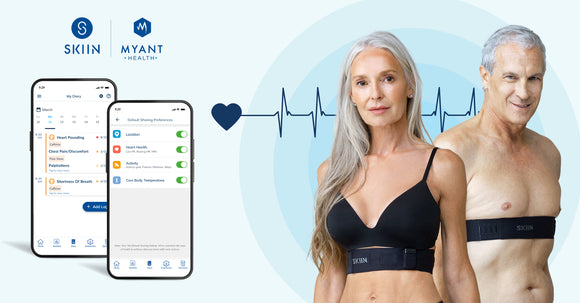
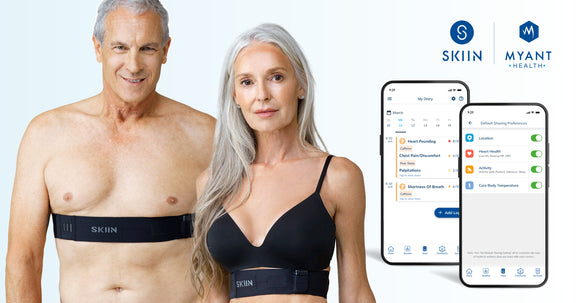
Take the right steps to a healthier life with Skiin
Written by Clarissa Pedrini Schuch, R&D Physiotherapist - Clinical Sciences and Curtis O’Connor, Content Writer Walking is one of the most popular forms of exercise, and for good reason. It’s easy, accessible, requires no special...